Drugs in India are in any case exorbitantly priced. And now come reports that the price authority may be wound up soon
In a country of 1.3 billion people, the government’s expenditure on healthcare rarely becomes a matter of pride. In 2013-14, India’s total healthcare expenditure was Rs 4.5 lakh crore, at about 4 percent of the GDP. But this figure was inclusive of all expenditure made by both public and private sectors. The government’s own health expenditure was merely 1.15 percent of the GDP, or in other words, just Rs 957 per person, according to the draft health policy of 2015. The 2016 budget had a healthcare allocation of 1.3 percent of the GDP. This, needless to say, is pathetically low.
Take a look at other nations: Thailand (3.2 percent), Brazil (4 percent), China (3 percent), and the US (8.3 percent), all have a higher proportion of public healthcare expenditure to GDP than India. To be fair, competing with the US may be a tall order, but India’s expenditure is way below the global mean of 5.4 percent.
Our government’s disinclination to spend on healthcare is reflected in the draft health policy 2015 as well. It says that India should aspire to spend 4.2 percent of its GDP on public health, while also suggesting that 2.5 percent would be more realistic.
In the midst of all this, the Indian pharmaceutical sector seems set to grow at a strong pace. As per a joint report by Assocham and TechSci Research, the industry would grow at an annualised rate of 8 percent. The market, as per the report’s estimates, would balloon to $55 billion in 2020 from the current levels of $18 billion. One can’t be faulted for feeling proud after reading these figures, but pride – proverbially – comes before a fall. And in this case, it would be the patients, not the industry, who will take the fall.
In the FY17 budget, the ministry of health and family welfare was allocated Rs 40,657 crore – Rs 463 crore short of the annual combined profits of the domestic pharmaceutical sector alone. In such a scenario, the actual burden of healthcare naturally gets transferred to individuals and households. This is reflected in India’s current health expenditure (CHE) of Rs 4.21 lakh crore, of which a massive 69 percent comes from people footing medical bills out of their own pockets, otherwise known as out-of-pocket (OOP) expenditures. Another dreadful fact is that 40 percent, or 1.68 lakh crore of the CHE, comprises money spent on drugs and medicines.
With expenditure on medicines this high, it becomes imperative that drug prices are kept under strict regulation so that interests of people come before those of pharmaceutical companies. However, the drug pricing policy in India is beset with the challenge of balancing corporate and public concerns. This is substantiated by a 2004 WHO report which finds that 67 percent of India’s population has no access to medicines. This is ironic considering that India is often called the ‘pharmacy of the world’.
There are more worrying signs on the horizon. Reportedly, the government is set to disband the National Pharmaceutical Pricing Authority (NPPA) – the regulatory authority that fixes and revises the price of controlled drugs in the country and penalises manufacturers in case of overcharging. Further, instead of bringing more medicines under the NPPA’s purview, the government is looking to cut down the list of price-controlled medicines. Surprisingly, this new offensive on public health is being unleashed by the Niti Aayog, which has written to the health ministry, calling on it to remove all “regulatory hurdles” from India’s pharmaceutical landscape, according to a report in the Economic Times.
Before we analyse the finer points on how drug regulation in the country went off track, it would be helpful to look at the evolution of the policy in India.
History of drug regulation
In the 1960s and 70s, prices of drugs in India were considered among the highest in the world. The governments of the day addressed this situation by introducing numerous price control orders in the 70s and the following decades. However, all these pricing orders progressively witnessed fewer drugs coming under their purview. For instance, in 1979, 347 drugs were placed under the pricing order. The list was then pruned to 142 under the 1986 order, with only 74 drugs remaining under the 1995 Drug (Prices Control) Order (DPCO). A policy directive released in 2002 sought to reduce the drugs under regulation to 35 but concerted civil society action ensured that the directive was challenged in the courts and stayed. While the 2002 policy directive remained suspended, the 1995 DPCO continued to operate. Meanwhile, the UPA government, working at snail’s pace, released a revised National List of Essential Medicines (NLEM) in 2011.
Thereafter, in May 2013, the centre notified the DPCO, whose Schedule I was the NLEM 2011. The drugs mentioned in this schedule came to be known as scheduled drugs and any formulation, that is, a medicine comprising one or more scheduled drugs came to be known as scheduled formulation. These drugs and formulations are subject to price control as mentioned in the DPCO 2013.
In 2015, the government started revising the NLEM and came out with a new list comprising of 376 drugs to be included in NLEM 2015. The expectation from the revised NLEM was that it would cover a larger share of the pharmaceutical market. However, the coverage was a dismal 18 percent, as per a pharmaceutical market research body, AIOCD Pharmasofttech AWACS. As per another estimation by PharmaTrac, through its January 2015 data, the coverage of price-controlled drugs further reduced to 13.4 percent.
Then there is the more important issue of pricing drugs, dominated by profit-oriented calculations in the market.
Cost-based vs market-based pricing
Unlike the current trend, the DPCOs of 1979, 1987 and 1995 were based on cost-based pricing method, which basically meant that a ceiling was placed on the profitability of the finished product. This was arrived at by calculating the cost incurred by the manufacturer in producing the drug and subsequently allowing him maximum allowable post-manufacturing expenses or MAPE. The DPCO 1995 provided a 100 percent MAPE margin. That is, if the cost of manufacturing a tablet is Rs 10, it could be sold for Rs 20.
However, with the DPCO 2013 coming into effect, market-based pricing (MBP) model was used. As per this model, ceiling prices are set on the basis of average retail prices of medicines sold by companies which have a market share of more than 1 percent. So, let’s say a drug for curing common fever is manufactured by 20 companies and 15 of them have a market share of more than 1 percent. Let us also assume that these 15 manufacturers have priced their drugs at more than Rs 10 per pill whereas the remaining five are selling the drug at a price less than Rs 10 per pill. In such a situation, the pricing of the drug under the DPCO will tend to fall on the higher side of the price spectrum.
Dr Sakhtivel Selvaraj, a senior public health specialist, tells Governance Now that ideal competition conditions exist only in about 25 percent of the Indian market, leaving the remaining space open to oligopolistic or monopolistic competition scenarios. In such market conditions, manufacturers are likely to quote higher prices for their finished products which means that the simple average of retail prices would also be on the higher side.
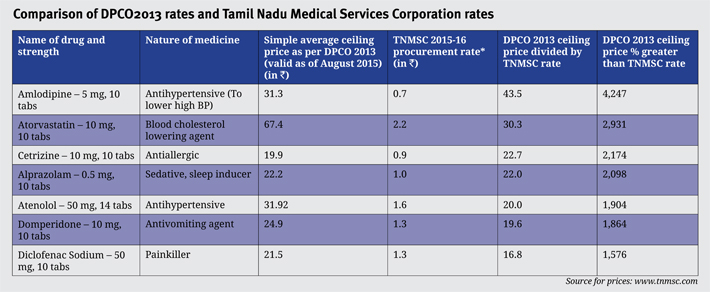
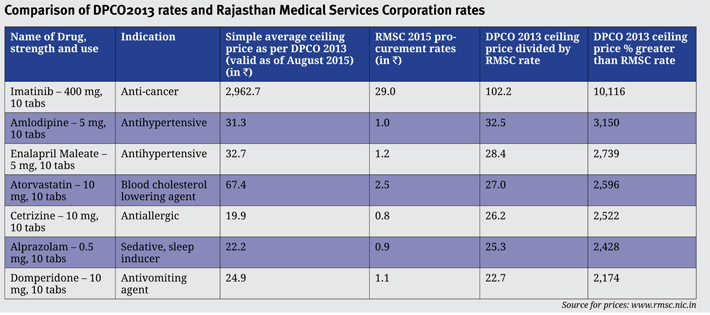
The exploitation of the patient is so blatant that private manufacturers often enjoy a pricing margin from 370 percent to even 4,247 percent, as compared to the prices at which state medical services corporation acquire drugs.
READ INTERVIEW: “In 50% of the Indian pharma sector, oligopolistic conditions prevail”
The escape route
The situation further aggravates as manufacturers have devised ingenious ways of escaping the DPCO. Citing the example of anti-diabetic drug Metformin, Dr Selvaraj explains that as the NLEM mentions only two dosages of Metformin, namely 250 mg and 500 mg, drug pricing restrictions would be applicable to only these two formulations and not the other formulations which manufacturers often produce, such as 200, 300, 400, 550 and 650 mgs.
Additionally, drug companies often expend large resources in persuading doctors and distributors to prescribe and supply only their own products. A 2014 study carried out by the Delhi School of Economics spoke to a practising surgeon, who shed light on the issue. The surgeon pointed out the ulterior “incentives” that motivate a number of prescriptions by doctors. He stated: “…one cannot ignore the role of what are euphemistically called marketing ‘incentives’, which basically mean the inappropriate influence pharmaceutical companies exert on doctors. This runs deep. Hospitals choose to stock only certain drugs in their in-house pharmacies and insist that hospitalised patients buy drugs only from the hospital pharmacy. Drug companies sell drugs to hospitals at a price much lower than what the patient is charged, further incentivising the hospital to stock their products. The cheaper brands often get left out in this game.”
Another drawback is the massive presence of fixed dose combination drugs (FDCs) in the domestic market. FDCs are combination of two or more active drugs consumed by the patient in a single dosage. While FDCs are useful in dealing with complex diseases like HIV/AIDS, tuberculosis and malaria, Indian manufacturers often resort to producing FDCs for regular ailments to escape the DPCO. They do this as the NLEM currently includes only 23 FDCs which make up for less than five percent of the Indian market. Drug producers often add an efficacious pharmaceutical ingredient with one that makes no significant therapeutic sense. This allows them a convenient ruse of wriggling out of the DPCO as they can claim that their particular combination of dose is not included in the NLEM.
India, as per Dr Selvaraj, has an FDC market share of 47 percent. In his opinion, no other country has such a massive presence of FDCs. Many of them don’t have any therapeutic efficacy and have also been declared dangerous by the Kokate committee, set up in 2014 to probe their efficacy.
To understand the kind of grasp FDCs exercise on the domestic market, have a look at a report released by AIOCD Pharmasofttech AWACS this year. It states that the industry stands to lose Rs 3,838 crore with the banning of 344 FDCs this year after the Kokate committee came out with its report. From another perspective, this is actually the amount Indian patients would save by not spending on inefficacious and irrational drugs. About 500 more FDCs are also reportedly under the scanner.
In September, the health ministry moved the supreme court seeking transfer of all the cases pending in different high courts before the apex court. Among those in the dock are major brands like Saridon, Corex, Benadryl and Glycodine. These FDCs, so ubiquitous in the market, are often sold at a premium compared to their non-branded counterparts.
While the situation looks bleak, all is not lost. Surprisingly, public procurement agencies have managed to beat the odds when it comes to battling the premium placed on drugs. Two corporations, one from Tamil Nadu and the other from Rajasthan, stand out for their competitive drug rates.
Showing the way
The Tamil Nadu Medical Services Corporation (TNMSC) was set up in 1994 in wake of a mega drug scam in the state. Learning from its mistakes, the state government introduced reforms in the drug delivery chain that resulted in a streamlined medical services corporation, procuring drugs for the entire state.
Prior to the incorporation of TNMSC, public hospitals were procuring drugs as per their own discretion, leading to massive variations in the quality and pricing of drugs, and also opening up prospects for illegal gratification. However, TNMSC, an autonomous body, not only eliminated these differences but also helped cut back on prices of drugs. For example, 10 strips of ciprofloxacin, an antibiotic for bacterial infections, were priced at Rs 525 during 1992-94. During 2002-03, the cost fell to Rs 88. Currently, they are priced at Rs 112.20; this still works out cheaper for patients as the current price is over four times less than what it was in the 90s.
Another state procurement agency that successfully controlled drug prices is the Rajasthan Medical Services Corporation. It, in fact, has been partly credited for contributing towards reducing poverty in the state. Families which often had to make catastrophic expenditure on healthcare, with major share incurred on medicines, got access to free medicines under the ‘Mukhyamantri Nishulk Dava Yojana’ launched in 2011. Under this scheme, 602 essential drugs, 73 surgical items and 77 sutures are provided without any charge. Additionally, the scheme has been aided by the government’s directive ordering state medical officers to prescribe drugs by their generic names. Prior to the launch of this scheme, around 44 lakh patients used to visit government hospitals per month. But after the scheme was introduced, the figure doubled to about 80 lakh. While this translates into greater spending by the state government, roughly of about Rs 300 crore per annum, it also means that OOP expenditure for people in Rajasthan has come down by Rs 2,500 crore.
The efficient performance of these public procurement bodies is evidence that despite an inclement pricing policy, it is possible to secure free or cheap medicines for the economically backward. The Modi government can take cue and revamp the drug policy to make sure that common and life-saving drugs reach a wider mass base at affordable rates which can, incidentally, have the effect of arresting poverty levels in the country.
However, the government seems to be motivated by the concerns of the pharmaceutical lobby. This is in stark contrast to the US government, which through its Department of Justice has stepped up its probe into suspected price collusion by a number of drug companies. News of the probe has sent share prices of Sun Pharma and its US arm Taro pharmaceutical in a free fall. A September report by IIFL, a financial services group, highlights how a number of drugs companies like Sun Pharma, Dr Reddy’s, Aurobindo, Lupin and Glenmark have increased prices of generic drugs anywhere from 150 to 800 percent on a year-on-year basis.
If the US can be so keen on cracking down on drug prices, why is India so eager on dismantling drug-pricing structures?
READ INTERVIEW: “There is power struggle as well as ideological struggle in the government”
(The story appears in the December 16-31, 2016 issue of Governance Now)

