India is mending rules and regulations for drug trials but loopholes remain concerning patient safety and transparency
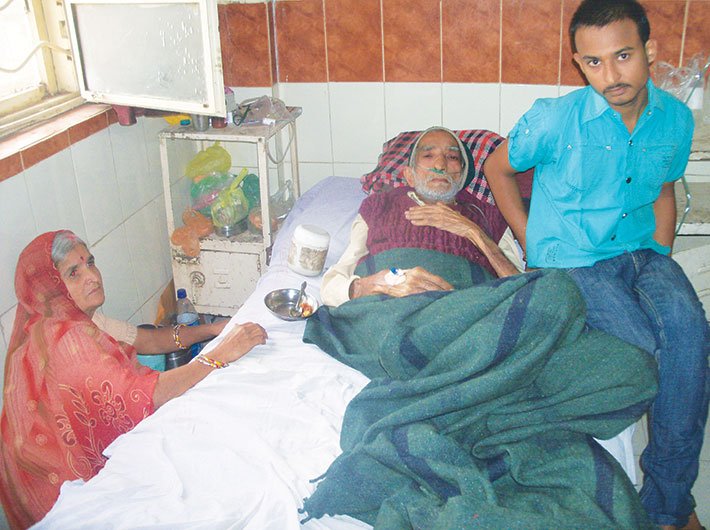
Pradeep Gehlot’s voice trembles even now when he talks of his father Srikrishna Gehlot’s death four years ago of a complication he developed in his chest.
“Father had only a mild chest pain and we took him to the Manorama Raje Chest Hospital in Indore. It is a government hospital and the doctor there immediately started the treatment. He made me sign some papers. In those days, I didn’t know much English and I was just 18 years old. So I didn’t understand what I was getting into. He told me that the treatment would be free of cost and they were using drugs made in Germany. The treatment went on for a year and then the doctor abruptly stopped it without giving reasons. Now my father was worse than ever. He could barely breathe. No other medicine could handle his condition and the German drug was not an option either. The doctor refused to treat him anymore. In a few months, he was dead,” he recalls.
Gehlot, an autorickshaw driver in Indore, blames the German drug for his father’s fate. He filed complaints with police and human rights bodies but he has never received any compensation or even acknowledgement of the wrong done to his father.
“How can we prove that he was a victim of drug trials?” he asks.
***
About 4,300 people have died after taking part in drug trials between 2005 and 2015. Only in 160 of these cases, the relatives have been given compensation. These are the figures that the Directorate of Health Services gave in reply to a Right to Information query in 2016. (If we consider 2005-16 data, 4,534 patients died but only 160 deaths were compensated. The compensation figures are only up to 2015, as the government could not give figures for 2016.)
In 2015, the union health ministry made drastic changes in the rules for conducting clinical trials, providing for compensation within three months of a death or an adverse event during a trial. The ministry made many other changes in the rules between 2013 and 2015 following a public interest litigation by an NGO in the supreme court in 2012. But have things become any better for the people who are subjected to trials as part of a process to test the efficacy of new drugs?
Now another petition has brought these changes before the supreme court, which will give a final ruling by early next year.
One of the changes the ministry has made, as mentioned before, has been ensuring compensation within three months. In 2016, 381 people died while undergoing clinical trials, according to the Directorate of Health Services reply to the RTI query. But just four persons were compensated as per the government reply. In November 2016, the reply said, no compensation had been paid for any of the 252 deaths during clinical trials. These cases were “under examination” even nine months later.
Another change made by the ministry concerns the ethics committees which monitor the trials. Every manufacturer planning to go for trial on a drug or a molecule must set up an ethics committee. Earlier, such committees were not even registered under any regime. Now, under the changed rules, these have to be registered.
“But how credible are these committees which are set up by those doing the trials?’’ asks Amulya Nidhi of the Swasthya Adhikar Manch, the NGO which has filed the petition in the apex court. “Shouldn’t ethics committees be institutional rather than private,” asks Nidhi.
Another change that has been brought about is in the provision for recording the consent by patients. Now, as per the rules, recording has to be done only for vulnerable sections. This again raises the question as to who is not vulnerable, especially when it concerns human health and life. These are questions that are raised only when one looks at the rules concerning clinical trials from the perspective of the safety of the subjects who undergo it.
As far as the industry and the researchers in the clinical trial field are concerned, the new regulations made in 2015 by the health ministry have received applause.
Chirag Trivedi, president of the Indian Society for Clinical Research (ISCR), recently said that following the new regulations, the ease of conducting trials and getting approvals has been fast-tracked and there would be a surge in clinical trials in 2018. There has been concern in the pharma industry about making rules stringent.
Data cited by ISCR shows that less than 1.5 percent of global clinical trials are being conducted in India. While there were 17 global clinical trials in 2013, the number went up to 87 in 2014, and dropped to 43 in the following year, reflecting the impact of the court case and the subsequent changes in rules.
The number of trials came down from 150 in 2014 to 110 in 2015 compared to over 300 trials approved in years before 2013. However, the 2015 regulations have been welcomed by the industry while the activists and patient bodies have expressed reservations about loopholes that remain.
The question remains as to how safe and how fair is the process of clinical trials as far as citizens subjected to it are concerned. If a person dies during clinical trials, what are the consequences for the investigators?
According to Amulya Nidhi, there is no law for punishing clinical trial violations. They just get barred from doing trials for a few months. “Does that equal the patient’s suffering?” asks Nidhi.
As per the regulations, the only compensation that a person suffering adverse events is daily payment of half the daily wage of an unskilled worker for a reasonable period.
So how safe is the patient undergoing clinical trial?
Pushkar Raj has been tracking clinical trials in Bikaner for the past one decade and says that the new rules are welcome but it is doubtful if they are being implemented.
The rules have laid down the amount of compensation for various types of outcomes of clinical trials, ranging from serious adverse events and disability to death.
The calculation of compensation for deaths is done using Rs 8 lakh as a base amount. For hospitalisation cases, hospital expenses are to be met and if the person is rendered disabled, then a sum is put in fixed deposit which will ensure monthly interest equivalent to half the minimum wage of an unskilled person.
Raj, whose organisation – Rashtriya Ekta Manav VIkas Sansthan – fights for the human rights of clinical trial victims, says that these formulae are on paper.
The data given out by the ministry confirms what he says. Only four out of 381 deaths in 2015 were compensated. No one knows how much these four people were paid as their details are not in public domain.
It is not just compensation amounts and the delays in compensation which are worrying. It is also the manner in which people are included in clinical trials without their knowledge.
Raj Verma cites the example of a man in his early fifties named Ashok, who ran a shop in Bikaner. He was admitted to the local government hospital following some illness. The head of the department said he had a heart problem and began to treat him with a medicine from a top pharma company.
A few months later he died as suddenly as he had fallen ill. The company sent Ashok’s family a cheque of Rs 2,50,000 after a year. Ashok’s wife says she had no clue why they were getting the money till she went to the hospital to donate a cooler there. The doctor informed her that the money was paid by the company whose medicines her husband had taken.
She and her son smelt something fishy and soon realised that they had been tricked into a clinical trial and Ashok’s death was probably linked to the medicine he took.
“They had no clue what to do,” says Pushkar Raj. Raj is still pursuing their case with the National Human Rights Commission.
Clinical Trials Registry of India (CTRI) set up by the ministry has been registering trials since 2007. However, its website does not give data on the number of trials going on or the number of subjects taking part. According to a study by the Department of Clinical Pharmacology, JIPMER, Puducherry, in 2011, about 400 trials are registered with CTRI each year and India does 2.9 percent of the total trials done globally with the US topping the list with 47 percent.
The top 10 diseases with maximum ongoing trials in India were found to be cancer, diabetes, coronary artery disease (CAD), asthma, chronic obstructive pulmonary disease (COPD), epilepsy, hypertension, schizophrenia, heart failure, stroke, HIV, and malaria. The study also showed that the number of clinical trials registered in infectious diseases like malaria and tuberculosis remain very low in India in spite of the disease burden.
Queries to the Drug Controller General by Governance Now elicited no reply on the matter of the number of total patients under trials.
So where does the common man go for information about the drug trials that are being conducted using people as research subjects? Some information is available to the common man on the website of the clinical trials registry of the United States!
It gives region-wise and country-wise breakup of trials that are on. As for deaths, the victims and relatives in the US can register on the death index and report any deaths caused by various reasons including trials.
In India the common man is still not in the picture and all data is with the bureaucracy. The supreme court is to consider the regulations made by the ministry and give its final judgment by next year. Will it finally herald a turning point for all concerned in clinical drug trials?
feedback@governancenow.com
(The article appears in the November 15, 2017 issue)